|
9/10/2023 0 Comments Atomic emission spectrum of helium
Bohr showed that this model gave quantitatively correct results by deriving the Rydberg Formula, which gives the wavelength of light emitted from the atom, again depending on the orbital transitions made by the electron (Bohr, 1913). 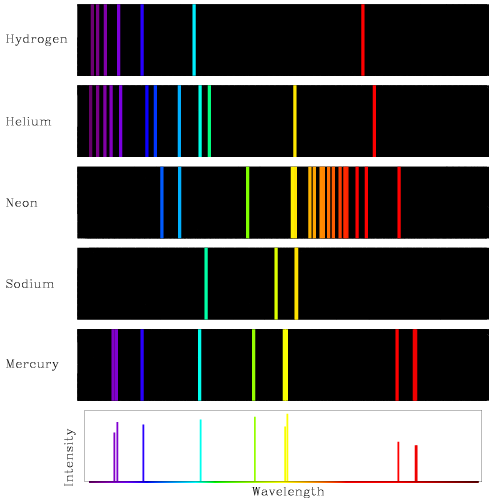
Energy would be released from the atom as light, and the wavelength of that light differed depending on the orbitals involved in the electron’s quantum transition. As energy was added to the atom, the electrons made transitions between orbitals. The Bohr model supposed that electrons orbit the nucleus in rigid orbitals or paths. In 1913, Niels Bohr introduced the Nobel Prize-winning quantized version of the Rutherford-Bohr model of hydrogen. Error between experimental and predicted ionization energies is about five percent, indicating that our model is reasonably accurate within the aforementioned parameters. We then predict the first and second ionization energies. 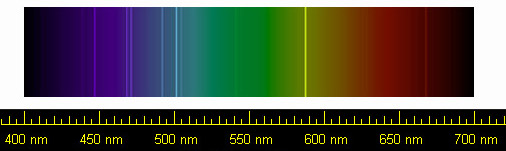
In this model we calculate an energy prediction for the unionized helium atom. The spectral lines calculated, namely the specific wavelengths of light emitted by elements when they are energized, correspond to an assumed possible scenario where the two electrons make orbital transitions in unison. We assume that the two electrons are at 180° apart in a circular orbit due to their mutual repulsion. We extend Bohr’s derivation, assuming a natural electron configuration, to explain some of the spectral lines of helium (He). Derived here is a simple model for helium and two-electron systems that may make some concepts, such as screening, easier for students to understand. Quantum and atomic physics can often be an abstract and difficult subject for students to learn.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
